The Availability Problem:
Imagine you have cancer, or chronic pain, or a progressive degenerative disease of some sort. You have exhausted the traditional treatment options available to you, and none of them have worked. However, there are treatments that are still undergoing clinical trials which might help you. They are not fully approved yet, but your situation is dire and you don’t have time to wait another 10 years for the trials to finish. Can you access those treatments?
In theory yes, you can access unapproved treatments through federal laws like the 2018 Right to Try bill, or through FDA pathways like “Expanded Access”. However these laws don’t mandate that the company making the drug gives it to you. And what you will find when you try to use your Right to Try, or Expanded Access, is that there are almost no treatments available for use.
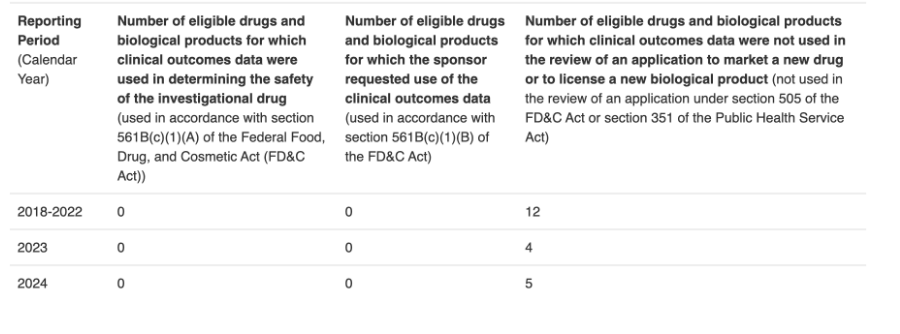
That’s why despite there being somewhere in the neighborhood of 13,000,000 Americans with terminal or serious illness, the FDA only grants about 2,000 Expanded Access requests per year, even though they approve 99% of all requests, typically within 24 hours. There just aren’t enough companies even bothering to apply.

No one really knows for sure how many patients have been treated under Federal Right to Try laws. What we do know is how many companies with drugs going through the clinical pipeline have used data from treating patients with RTT, when submitting data for their treatments to the FDA. Even if we assume the usage numbers are 2-3X higher, they are still pretty abyssmal.
Risk and Reward:
But what is it about these pathways that makes companies avoid them like the plague? The answer is three words: Terrible Risk/Reward.
The companies who own the rights to the treatment you want to access are called “Biotechs”. Now Biotechs are not like the big pharma companies you think of, they don’t make billions of dollars. In fact, they usually don’t make any money at all. They’re the small companies that do the research to get the drug approved. This approval process costs somewhere in the neighborhood of 1-2 billion dollars, and takes somewhere in the neighborhood of 10 years.
Biotechs can’t sell their drug pre-approval, so the only way they can fund this process is by raising money. But they don’t get this money all at once. It’s given to them one chunk at a time, as investors say “Okay do this first round of trials, if it goes well we might fund the next one”. This means that just to stay alive as a company and keep the treatment moving through the process, Biotechs must be able to raise money.
And competition is fierce, there’s a lot of Biotechs competing over a small pool of capital. So to raise successfully, you need to be absolutely flawless. As a result of this the executives who run Biotechs are extremely risk averse. They will inherently stay away from anything that has even a slight chance of jeopardizing their clinical trial.
So what does this have to do with Right to Try? Well, treating you under Right to Try is a risk. If something goes wrong, there's the risk of a lawsuit, there's the risk of other investors whose capital is needed to keep the company alive finding out about the bad outcome, which leads to the biotech not being able to raise the next round which it needs to survive. There's also some risk the FDA will come down on you, though they really only do this in extreme circumstances.
So for a Biotech, treating patients before their drug is approved is enormously high risk. That said, these are humans at the end of the day and typically run by founders that went into medical sciences because they are motivated to save lives. If they can justify the risk to their board, many of them would take it? So what’s the reward for participating in Right to Try?
Nothing. There is no reward. Actually, the Biotech will probably lose money treating you.
Federal Right to Try and Expanded Access both only allow the Biotech to charge “at cost”, basically meaning they can recoup the cost of the drug. There’s a ton of other costs that they will probably not be able to get reimbursed for. The lawyer who had to be hired to check that the company could do this, the hours that the Board spent drawing up plans and debating whether or not to do it, the time someone needed to take to engage the FDA and answer your doctor’s questions, and more. All of that is probably money lost.
So the pathways you want to use only work if there is a company willing to basically risk their reputation, and even their company’s survival, just for the chance to lose money treating you. And that’s why no one uses them.
State Laws Are Addressing The R/R Problem:
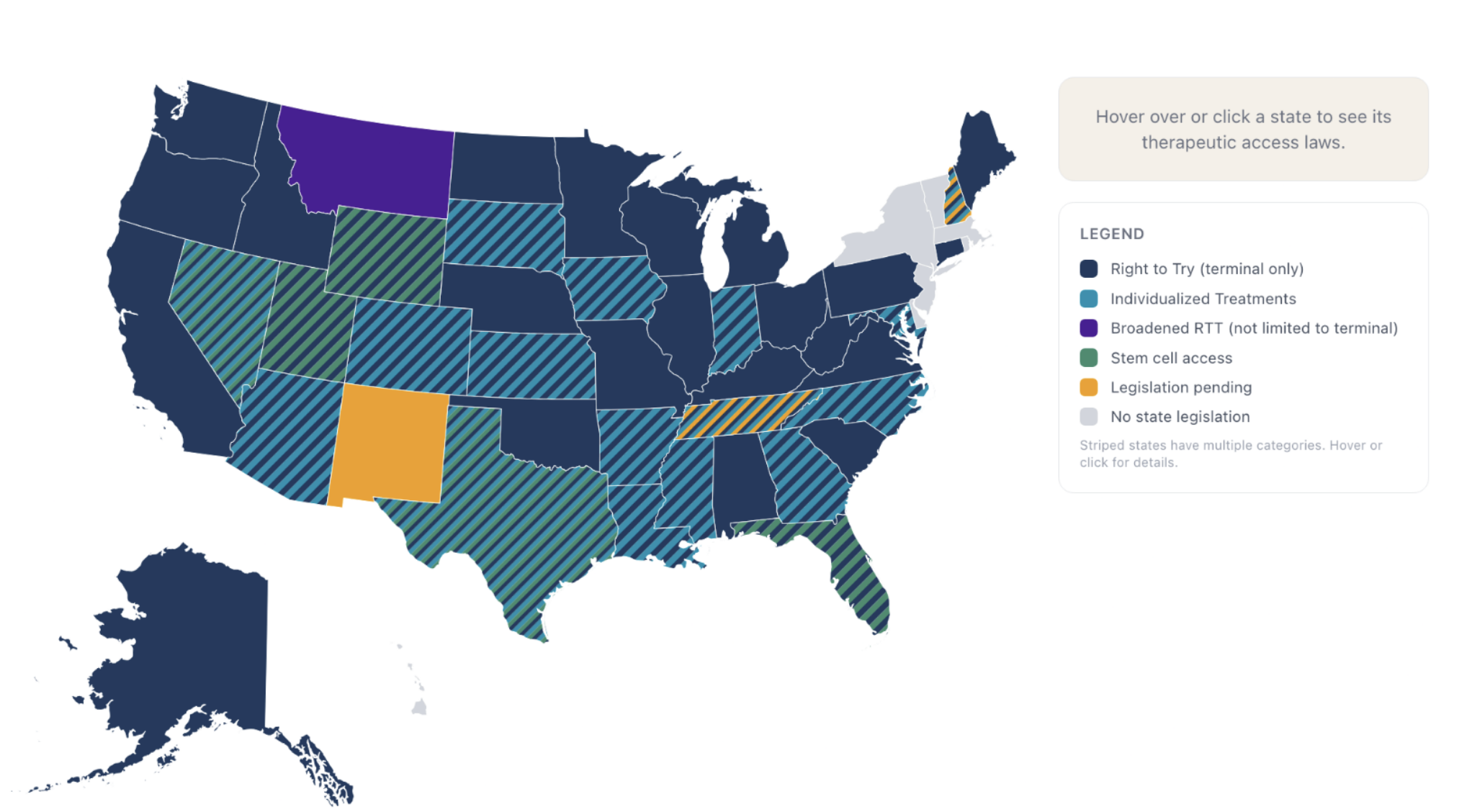
In 2025 Montana passed an expanded Right to Try bill, SB 535. This bill was specifically crafted to fix the risk/reward problem from earlier. Among other things, SB535 allows for Biotechs to make a profit for treating you instead of taking a guaranteed loss. The idea behind this was that by fixing the Risk/Reward, Montana could attract companies that wouldn’t otherwise make their treatment available.
And it has worked.
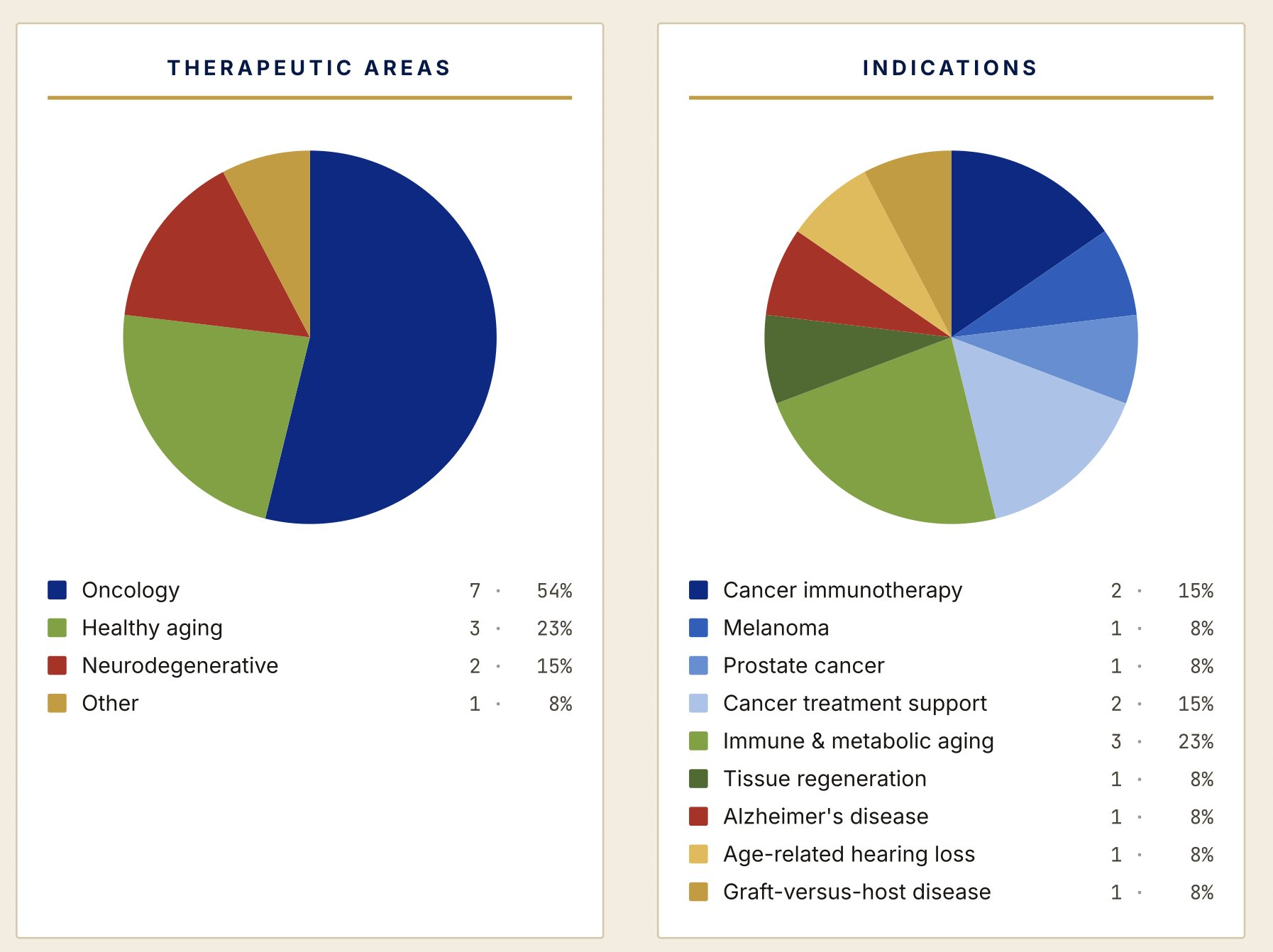
I work sourcing treatments for use under SB535. I am paid to talk to Biotechs whose treatment is not available, and convince them to come to Montana. Patients should start receiving treatment some time in June or July, we may very well have more treatment options available in Montana within the first year than have been used under Federal Right to Try in the last 8 years combined.
Here are two charts showing what will be available through just us in Montana basically as soon as the program launches:
And Montana is not alone in this. New Hampshire has HB701, a similar law but terminal illness only.
However, once again, there is a risk of these laws only working "on paper".
Somehow it's the FDA, Again:
Remember those biotechs I mentioned? Well, whether or not some state law says they can come treat patients, the FDA still has the ultimate say. The FDA could shut down a biotech's clinical trial for coming to Montana, or New Hampshire. This is a power they reserve unto themselves.
As a result, all of the treatments I just mentioned and many more, will only condition their participation in state programs like Montana and New Hampshire's on receiving Safe Harbor. Basically this is where the FDA exercises something called "Enforcement Discretion", and in doing so promises the biotechs that they won't get in trouble for coming to treat patients under state law.
Without this, as nice as these state laws are, only the most risk tolerant providers around will participate. This means there will be some treatments available, but only something like 10% of what the numbers could be, and it means patients will continue to suffer and die from lack of access.
Convincing people in the House and Senate to back up efforts to get the FDA to promise enforcement discretion is what I've spent the last 6 or so months working on.
This actually matters a lot for states other than Montana and New Hampshire.
In Utah, there is a stem cell access bill. There is also a great company called Discgenics making stem cell based therapies which are in late stage clinical trials based in Salt Lake City. You cannot access Discgenics treatment under Utah state law. That's pretty ridiculous! Florida, Texas, New Hampshire, and maybe soon Tennessee, have similar laws. So you can get off label stem cells, but nothing with actual clinical trials backing them.
In 17 states (AZ, NV, LA, MD, MI, NC, AK, TN, KS, SD, IN, GA, CO, IA, TX, NH, and WV) there are Right to Try 2.0 laws, also known as Right to Personalized/Individualized treatment laws. These are laws which allow for you to access treatments based on your personal genetic code, a class of treatments which is widely believed to be the silver bullet for many rare diseases and cancers. Like the other laws I have mentioned, any company that participates in one of these does so at their own risk.
Discuss